Podiatrists are highly trained to diagnose and treat problems relating to the feet and lower legs. Their extensive training includes undergraduate studies and 4 years of podiatric medical school.
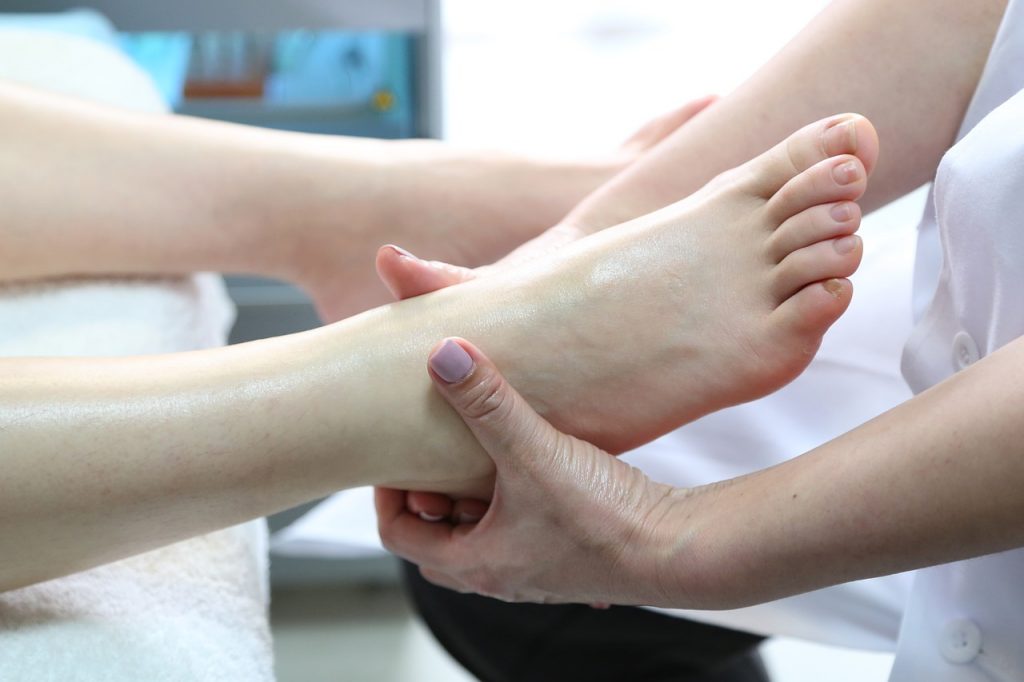
Foot massages relieve tight muscles by increasing muscle temperature, reducing restriction, and improving tissue elasticity. They also stimulate the lymphatic system, removing waste products from injured muscles more effectively.
Deep Relaxation
A foot massage stimulates certain nerve endings in the feet and releases endorphins, which are known as the body’s natural “feel-good” hormones. Combined with aromatherapy, it can help to reduce stress and encourage an overall state of relaxation.
Aside from reducing stress, foot massage has been shown to be an effective treatment for high blood pressure. Regular foot massages have been found to lower both systolic and diastolic blood pressure levels in individuals with hypertension, and can also help to lower blood triglyceride levels.
Another benefit of foot massage is its ability to alleviate headaches and migraines. Studies have found that massaging the reflex point on the top of the foot, located between the big toe and second toe, can relieve tension in the head and neck, thereby alleviating these symptoms.
Foot massage is also known to improve and support the immune system. Stimulating the foot’s lymph fluid circulation during a massage can increase white blood cell activity in the body, which is important for fighting infections and strengthening the immune system.
In addition to promoting relaxation and relieving stress, foot massage can also improve sleep quality. A lack of sleep can lead to a number of health problems, including fatigue and depression. Research has found that foot massage can significantly improve the quality of a person’s sleep by decreasing the levels of cortisol, which is a stress hormone.
Increased Healing
Remedial massage is a great way to increase the healing process and help muscles that are injured. Remedial massage increases relaxation and improves muscle condition through manipulation of the superficial and deep muscles. Relaxed muscles promote greater healing and decrease pain, restriction, and inflammation.
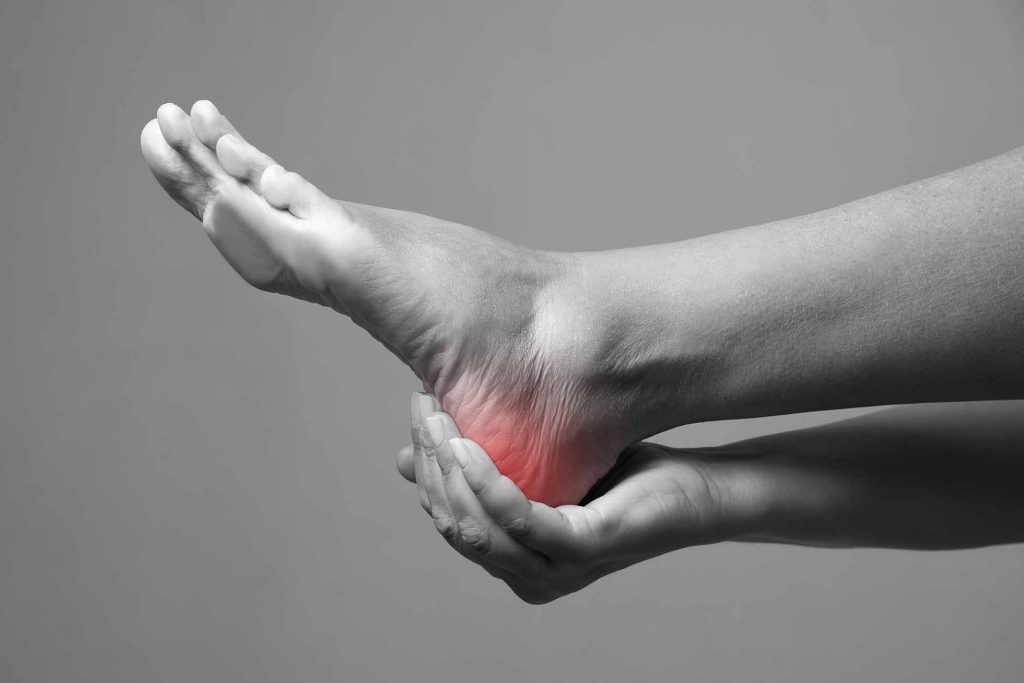
Foot massage encourages blood flow and lymphatic drainage reducing excess fluids that cause swelling. This allows tissue to return to a normal state of contraction and movement. This can be very beneficial for people with conditions such as edema, lymphoedema, and chronic heel pain.
A massage also helps to break down lactic acid buildup and encourages the removal of toxins. This is particularly helpful for people with conditions such as shin splints, plantar fasciitis, and Achilles tendonitis. It also can be a very effective form of treatment for Sever’s disease.
Unlike a regular foot massage, reflexology involves manipulating specific areas of the feet called “reflex points” which correspond to parts of the body. Kneading the soft fleshy ball of the foot, pulling on the toes, tracing around the heel, and pushing down into the arch are all part of this therapeutic treatment. Foot reflexology has been shown to treat a wide range of conditions including stress and anxiety, headaches, migraines, back pain, and more. It has even been known to relieve cancer-related pain.
Reduced Swelling
Swelling in the ankles and feet can be a frustrating condition to live with. Whether you’re dealing with it from long days on your feet, hours of travel, surgery, or pregnancy, the swelling can feel uncomfortable and restrict movement. But you can relieve swelling from everyday causes with a few simple steps. These include drinking plenty of water, limiting sodium and salt intake, increasing your activity level, and using diuretic supplements or lymphatic drainage massage.
Foot massages are a great way to relieve fluid retention because they help promote circulation and reduce inflammation. They can also be used to stimulate reflex points that aid in the body’s natural healing processes. Foot massage is especially beneficial for those with diabetes and hypertension, as it can improve their blood flow.
While most cases of swelling will subside on their own, it’s important to get a medical assessment if your swelling doesn’t go away or is accompanied by shortness of breath, chest pain, or pressure in the chest. If you have an underlying medical condition, such as heart failure or kidney disease, your doctor may recommend medical treatment to prevent further damage. Podiatrists at Modern Medicine can perform a range of treatments, including MLD, to help reduce edema and speed up the healing process. They can even use specific techniques, such as arch rubs and toe bends, to help you manage your symptoms at home.
Treating Scars
Often, scar tissue can cause restrictions in the foot’s mobility. This can lead to a loss of movement of the foot, increased pain, and discomfort. Remedial massage can help to soften scars, break down adhesions, improve circulation, and boost the body’s natural healing process.
Athletes and people with foot problems often have noticeable scars, as do people who have had surgery on the feet for conditions such as bunions, hammer toes, and fractures. These scars can make it difficult for the foot to move freely and if they rub against footwear or other surfaces can reopen them, slowing the healing process and making them more visible.
Remedial massage is a hands-on therapy that involves the use of techniques including deep tissue, friction, and kneading to treat muscle and joint dysfunctions. This style of massage is most effective for treating injuries or chronic aches and pains.
Your remedial massage therapist will start your appointment by asking you about your health history and why you are seeking treatment. They will then observe your posture, and flexibility and do a few range of motion tests to gain more insight into your injury or symptoms. They may also suggest dry needling (trigger point therapy) if they think it would be beneficial to your treatment. This is a great addition to any massage and can be used for many different types of injuries.
The combination of remedial massage and podiatry offers a comprehensive approach to foot wellness. From promoting deep relaxation and reducing stress to enhancing healing processes and addressing specific conditions, these therapies provide holistic benefits for foot health. Whether it’s relieving tight muscles, reducing swelling, treating scars, or improving circulation, the expertise of podiatrists and the therapeutic techniques of massage therapists contribute to maximizing foot well-being. Embracing these practices not only alleviates immediate discomfort but also supports long-term mobility, functionality, and overall quality of life.



 Many people ask the question, “Do chiropractors fix injuries?” In my experience, the answer is an obvious “yes”. At chiropractic clifton hill, we treat injured athletes and patients. I have even treated injured boxers who were out of action because they badly injured their backs or other physical injuries that required immediate surgery. I have also treated patients and athletes with musculoskeletal problems such as herniated discs, spinal stenosis, and arthritis.
Many people ask the question, “Do chiropractors fix injuries?” In my experience, the answer is an obvious “yes”. At chiropractic clifton hill, we treat injured athletes and patients. I have even treated injured boxers who were out of action because they badly injured their backs or other physical injuries that required immediate surgery. I have also treated patients and athletes with musculoskeletal problems such as herniated discs, spinal stenosis, and arthritis.

 At some point in life we all end up running across a physical problem that infringes on living the life we want – a pain, persistent ache, headache,
At some point in life we all end up running across a physical problem that infringes on living the life we want – a pain, persistent ache, headache, 

 Is Your Child Suffering from Stuttering?
Is Your Child Suffering from Stuttering?
 When I started as a police officer, the training program included a drug-testing policy that was implemented after an outbreak of drug crimes in my department. Prior to that, my officers were only required to perform drug screening at the scene of drug crimes and at
When I started as a police officer, the training program included a drug-testing policy that was implemented after an outbreak of drug crimes in my department. Prior to that, my officers were only required to perform drug screening at the scene of drug crimes and at 


 A recent study has shown that the role of healthcare professionals in improving healthcare quality is increasingly being recognised as a key driver of improved patient care. As the UK aims to move towards more sustainable healthcare practices, professionals are recognising the need to have a broader understanding of how they can contribute to this improvement. The role of healthcare professionals is not always easily understood by patients and therefore they are often at a loss to explain their work and what they do. It is clear that this leads to confusion about how they should act and contribute to patient care.
A recent study has shown that the role of healthcare professionals in improving healthcare quality is increasingly being recognised as a key driver of improved patient care. As the UK aims to move towards more sustainable healthcare practices, professionals are recognising the need to have a broader understanding of how they can contribute to this improvement. The role of healthcare professionals is not always easily understood by patients and therefore they are often at a loss to explain their work and what they do. It is clear that this leads to confusion about how they should act and contribute to patient care.

